 
Fourier transform methods are often used to enhance the sensitivity of infrared methods, and there are some specialized IR techniques that are used as well. IR bands are narrower than observed in UV/VIS spectra so instrumental deviations to Beer’s Law (e.g., polychromatic radiation) are of more concern. Also, IR detectors are much less sensitive than those for the UV/VIS region of the spectrum. One limitation to the use of IR spectroscopy for quantitative analysis is that IR sources have weak power that enhances the noise relative to signal and reduces the sensitivity of the method relative to UV/Visible absorption spectroscopy. In OCS, the oxygen atom is more electronegative than the carbon atom, and the sulfur atom is less. The larger the difference in electronegativities of bonded atoms, the larger the. The dipole moment of chloromethane, 12.9 D, is higher than that of dichloromethane, 9. This means that CO2 is nonpolar and has no dipole moment. 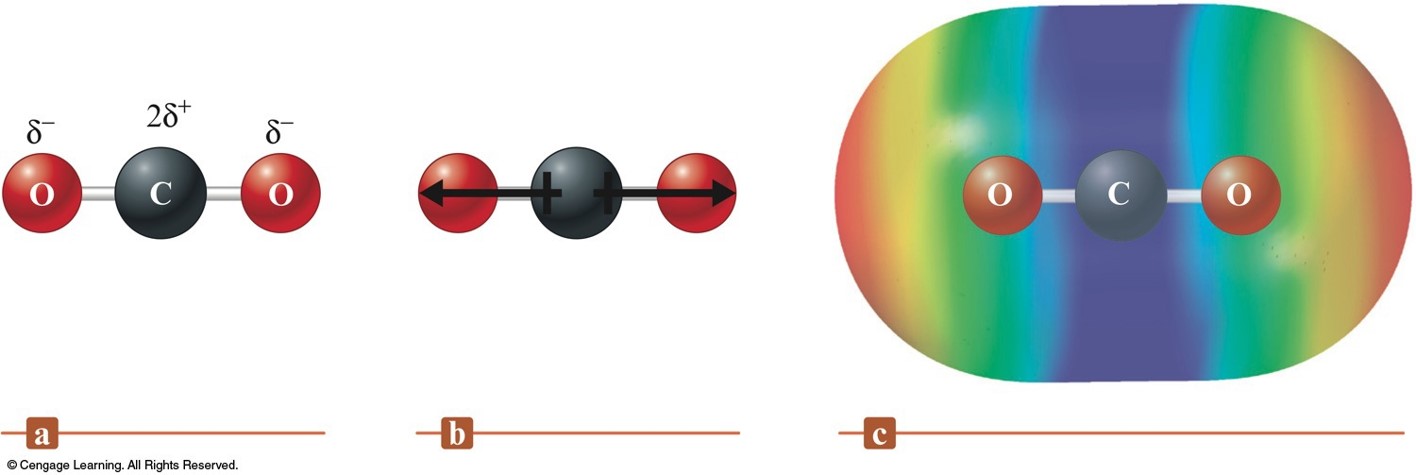
Dipole moments tell us about the charge separation in a molecule. The total number of valence electrons in the CH2Cl2 molecule is 20. It proves that CH2Cl2 is polar but a moderate polar as the difference between their electronegativity is quite small. IR spectroscopy can also be used for quantitative analysis. Dipole moment ( ) is the measure of net molecular polarity, which is the magnitude of the charge Q at either end of the molecular dipole times the distance r between the charges. So, electronegativity difference between C-H0.3 and C-Cl0.6. When using IR spectra for compound identification, usually a computer is used to compare the spectrum of the unknown compound to a library of spectra of known compounds to find the best match. Therefore, while each compound has a unique IR spectrum (suggesting that IR spectroscopy ought to be especially useful for the qualitative analysis – compound identification – of compounds), interpreting IR spectra is not an easy process. IR spectra are recorded in reciprocal wavenumbers (cm -1) and there are certain parts of the mid-IR spectrum that correspond to specific vibrational modes of organic compounds.Īn important consideration is that as molecules get complex, the various vibrational modes get coupled together and the infrared (IR) absorption spectrum becomes quite complex and difficult to accurately determine. Parellel lines in the well represent vibrational energy levels. Potential energy well for a covalent bond. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
